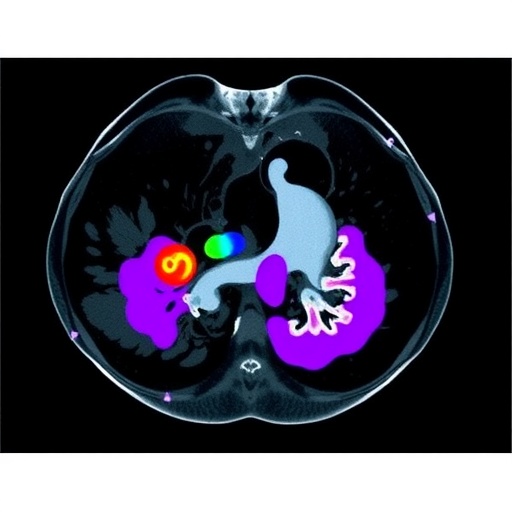
In a significant advancement for medical imaging and artificial intelligence, researchers have introduced BUSGen, a pioneering foundation generative model specifically designed for breast ultrasound analysis. Trained on a comprehensive dataset of over 3.5 million breast ultrasound images, BUSGen aims to transform the early detection, diagnosis, and prognosis of breast cancer, addressing challenges in a domain where interpretation has historically been complex for both radiologists and computational models.
The intricacies involved in breast ultrasound imaging—the complex anatomy, varied pathological manifestations, and inconsistencies in image acquisition—have long posed hurdles in accurate assessment. BUSGen employs innovative foundation generative modeling techniques that capture essential clinical knowledge, resulting in the generation of highly realistic and informative synthetic images. This extensive training allows BUSGen to understand breast tissue structures and identify deviations that may indicate malignancies, thus enhancing its applicability across numerous diagnostic tasks.
A standout feature of BUSGen is its few-shot adaptation capability. Unlike traditional models that necessitate extensive retraining with new datasets, BUSGen can swiftly adapt to different tasks using minimal examples, enabling the creation of precise synthetic datasets tailored to specific clinical inquiries. This approach is particularly beneficial given the challenges associated with gathering and annotating large medical datasets, which are often hampered by costs and privacy concerns.
Moreover, the synthetic data produced by BUSGen facilitates data augmentation, addressing the issue of class imbalance that often plagues medical imaging, where examples of pathological cases are limited. Models powered by BUSGen-generated data have shown enhanced performance compared to those trained solely on real patient data. Notably, in breast cancer diagnosis, BUSGen-derived models have surpassed existing state-of-the-art models, demonstrating superior diagnostic accuracy in capturing subtle features that might be overlooked by human observers.
BUSGen’s efficacy is underscored by its performance in comparative evaluations against expert radiologists. The model surpassed all nine board-certified radiologists involved in early diagnosis assessments, achieving an average sensitivity improvement of 16.5% with a highly significant p-value (<0.0001). This remarkable performance indicates BUSGen’s potential to enhance clinical practice, reducing the risk of missed diagnoses and enabling timely interventions in patient care.
Beyond improving diagnostic accuracy, BUSGen addresses ethical considerations surrounding medical data sharing. The synthetic datasets it produces are designed to maintain both statistical and pathological integrity while ensuring complete de-identification of patient information. This capability is transformative for collaborative research, as it allows for the sharing of rich datasets without compromising patient confidentiality, thereby accelerating innovation in the field of breast ultrasound AI.
The architecture of BUSGen reflects advancements in deep learning, integrating generative model strengths—potentially leveraging methods like Generative Adversarial Networks (GANs) or diffusion models—with specialized knowledge derived from extensive breast ultrasound collections. This integration enables the authentic synthesis of ultrasound images, a feat complicated by the inherent noise and artifacts typical of ultrasound imaging. The successful implementation of BUSGen highlights the meticulous engineering required to harmonize generative diversity with clinical accuracy.
In addition to its diagnostic capabilities, BUSGen supports prognosis prediction by identifying subtle imaging biomarkers indicative of disease progression. This aspect enhances personalized medicine, allowing physicians to tailor treatment plans based on individual tumor characteristics and expected disease trajectories, which could ultimately improve patient survival rates and quality of life.
The researchers have also examined how scaling synthetic data affects training workflows, revealing that increased volumes of generated data, when combined with real patient images, lead to continual improvements in model performance. This insight is pivotal for guiding future strategies in resource allocation and dataset construction, further emphasizing the synergy between real and synthetic data in advancing AI-driven solutions in medical imaging.
BUSGen represents a significant step toward democratizing advanced AI tools for breast ultrasound analysis, particularly in healthcare settings with limited resources. By generating adaptable, task-specific datasets, BUSGen lowers barriers for institutions that may lack extensive annotated images or robust computing infrastructure. This democratization is vital for addressing inequities in breast cancer outcomes globally, especially in regions where early detection capabilities are still evolving.
The enthusiasm surrounding BUSGen within the scientific community illustrates the potential for foundational models combined with domain-specific generative capabilities to reshape medical AI landscapes. Its successful development may inspire further research into generative models tailored for different imaging modalities and diseases, potentially igniting a new era of innovation driven by synthetic data in healthcare.
Looking ahead, the integration of BUSGen into clinical workflows could bolster radiologists’ capabilities by providing high-quality, annotated synthetic data for ongoing learning and refinement. Clinical decision support systems that utilize BUSGen-generated insights could expedite the identification of suspicious cases, prioritize urgent evaluations, and minimize diagnostic variability. Such advancements underscore the necessity for rigorous validation and ethical oversight to maintain patient trust and uphold safety standards.
In conclusion, BUSGen signifies a critical advancement in breast cancer imaging, merging foundational AI strengths with clinical expertise to enable breakthroughs in early detection and personalized care. As the incidence of breast cancer continues to rise globally, innovations like BUSGen provide a beacon of hope, offering tangible solutions for smarter, faster, and more equitable diagnoses in women’s health.
See also Meta Launches Spark Muse AI Model, Competes with OpenAI and Anthropic on Key Benchmarks
Meta Launches Spark Muse AI Model, Competes with OpenAI and Anthropic on Key Benchmarks Meta Launches Muse Spark, Outperforming GPT-5.4 in Health AI Benchmark by 2%
Meta Launches Muse Spark, Outperforming GPT-5.4 in Health AI Benchmark by 2% OpenAI Shares User Data Policy: Questions Retained for 30 Days Under Privacy Principles
OpenAI Shares User Data Policy: Questions Retained for 30 Days Under Privacy Principles Meta Launches Muse Spark AI Model with Multimodal Reasoning and Multi-Agent Support
Meta Launches Muse Spark AI Model with Multimodal Reasoning and Multi-Agent Support